
Empowering Individuals
Supporting Families

Registration is now OPEN for the 2025 International PWS Conference. Join PWSA | USA, IPWSO, and FPWR June 24-28, 2025, in Phoenix, Arizona!
Registration is now open for the 2025 International Prader-Willi Syndrome Conference in Phoenix Arizona! Prader-Willi Syndrome Association USA, FPWR and IPWSO are coming together to bring you the most up-to-date medical information, help and HOPE for parents and caregivers, a safe and fun space for your loved ones with PWS and their siblings, and a time to connect with other families. Space at the camps is limited; once they are full, we will close registration. Mark your calendars and register early! Find additional details and the Conference and Camps registration form at the button below.

Between July 1 – August 15, 2024, PWSA | USA is collecting artwork for Theme #2 in our PWS Rare Aware Art Share! We’re looking for artwork that answers the question:
What's something unique about where you live?
Think about where you live. Look out your window or take a walk in your neighborhood or town. Describe what you see in your art piece. Show us what your world looks like. If you can, include yourself in your artwork. How do you fit into the world you live in?
Prader-Willi Syndrome Association | USA
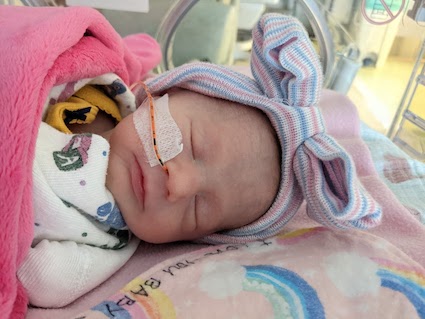
New Diagnosis?
Fill a New Diagnosis form to receive our Package of Hope, a Parent’s Guide to Prader-Willi Syndrome.
Click Here

Join our Newsletter
Join our mailing list for Prader-Willi Syndrome Association updates and other relevant information.
Sign Up

Update Contact Info
Update your contact information to stay informed about all the latest news and events from PWSA USA.
Update
What we do

PWS Advocacy & Awareness
Increasing awareness and effectively advocating for Prader-Willi syndrome at the state and federal level is a critical component of our mission. We seek to involve all members of our community and work to keep you informed on the best ways to affect change.
Learn about Prader-Willi Syndrome (PWS)



PWS Family Support
PWSA | USA' Family Support team provides individuals diagnosed with Prader-Willi syndrome, their families, and care providers with critical information and resources on PWS. We also provide education to medical providers, schools, and professional care givers through ongoing trainings, toolkits and other valuable resources. We support the family from the NICU through all stages of the PWS journey.

Prader-Willi Syndrome Research
PWSA | USA seeks to support research projects with the potential for immediate and high impact for the Prader-Willi Syndrome community. The goal is to fast-track better treatment for the syndrome, and we proudly collaborate with partners representing pharmaceutical companies, research universities, and more to achieve that goal.


PWS Events & Fundraisers
Upcoming Events
REGISTER IN ADVANCE HERE Presenters: Lynn Garrick, RN, MSN, PWSA l USA Medical and Research Coordinator, Barb Dorn, RN BSN, PWSA l USA Volunteer Overview: This webinar will highlight current research in the area of aging and PWS. It will also identify the most common health issues being reported by parents, guardians and caregivers. The […]

REGISTER IN ADVANCE HERE Presenters: Barb Dorn, RN BSN, PWSA l USA Volunteer Overview: This webinar will continue discussion on commonly reported health issues by sharing risk factors, screening and monitoring tools as well as prevention and management strategies for additional health topics – respiratory issues, weight gain/obesity, low bone density, cardiac issues (high BP, […]

Join PWSA | USA for our August ECHO 4 PWS Webinar, Cardiac Concerns with Dr. James Loker. On August 15, 2024, at 5:00 p.m. EST, Dr. Loker will discuss Cardiac disease, which accounts for 16% of all deaths in PWS. He will share the unique features of PWS that may predispose them to cardiovascular problems. […]

Visit our Blog
Growing up Rare
contributed by Justice Rickenbach My name is Justice Faith, and I am 20 years old. I have: Prader-Willi syndrome Narcolepsy…
PWS Aging Research and Health Update Webinar Series: Comprehensive Insights and Strategies for Caregivers
Join us for the PWS – Aging Research and Health Update Webinar series, featuring Lynn Garrick, RN, MSN, and Barb…
Soleno Therapeutics Submits New Drug Application to FDA for PWS Treatment
On June 28, 2024, Soleno Therapeutics announced the company officially submitted a New Drug Application (NDA) to the U.S. Food…
Lifestyle Practices to Manage Gastroparesis
The lifestyle aspect of managing gastroparesis is often overlooked or underestimated. Some people think it is too simple a…
Creating a Parent Input Statement for Your Child’s IEP
Contributed by PWSA | USA’s Director of Family Support Stacy Ward, MS As a parent, you know your child best…
Soleno Therapeutics Reports Positive Results from DCCR Study C602 for Prader-Willi Syndrome
Soleno Therapeutics, Inc. has revealed positive outcomes from the randomized withdrawal phase of Study C602, an extended treatment study of…







 Jennifer Bolander has been serving as a Special Education Specialist for PWSA (USA) since October of 2015. She is a graduate of John Carroll University and lives in Ohio with her husband Brad and daughters Kate (17), and Sophia (13) who was born with PWS.
Jennifer Bolander has been serving as a Special Education Specialist for PWSA (USA) since October of 2015. She is a graduate of John Carroll University and lives in Ohio with her husband Brad and daughters Kate (17), and Sophia (13) who was born with PWS. Perry A. Zirkel has written more than 1,500 publications on various aspects of school law, with an emphasis on legal issues in special education. He writes a regular column for NAESP’s Principal magazine and NASP’s Communiqué newsletter, and he did so previously for Phi Delta Kappan and Teaching Exceptional Children.
Perry A. Zirkel has written more than 1,500 publications on various aspects of school law, with an emphasis on legal issues in special education. He writes a regular column for NAESP’s Principal magazine and NASP’s Communiqué newsletter, and he did so previously for Phi Delta Kappan and Teaching Exceptional Children. Evan has worked with the Prader-Willi Syndrome Association (USA) since 2007 primarily as a Crisis Intervention and Family Support Counselor. Evans works with parents and schools to foster strong collaborative relationships and appropriate educational environments for students with PWS.
Evan has worked with the Prader-Willi Syndrome Association (USA) since 2007 primarily as a Crisis Intervention and Family Support Counselor. Evans works with parents and schools to foster strong collaborative relationships and appropriate educational environments for students with PWS. Dr. Amy McTighe is the PWS Program Manager and Inpatient Teacher at the Center for Prader-Willi Syndrome at the Children’s Institute of Pittsburgh. She graduated from Duquesne University receiving her Bachelor’s and Master’s degree in Education with a focus on elementary education, special education, and language arts.
Dr. Amy McTighe is the PWS Program Manager and Inpatient Teacher at the Center for Prader-Willi Syndrome at the Children’s Institute of Pittsburgh. She graduated from Duquesne University receiving her Bachelor’s and Master’s degree in Education with a focus on elementary education, special education, and language arts. Staci Zimmerman works for Prader-Willi Syndrome Association of Colorado as an Individualized Education Program (IEP) consultant. Staci collaborates with the PWS multi-disciplinary clinic at the Children’s Hospital in Denver supporting families and school districts around the United States with their child’s Individual Educational Plan.
Staci Zimmerman works for Prader-Willi Syndrome Association of Colorado as an Individualized Education Program (IEP) consultant. Staci collaborates with the PWS multi-disciplinary clinic at the Children’s Hospital in Denver supporting families and school districts around the United States with their child’s Individual Educational Plan. Founded in 2001, SDLC is a non-profit legal services organization dedicated to protecting and advancing the legal rights of people with disabilities throughout the South. It partners with the Southern Poverty Law Center, Protection and Advocacy (P&A) programs, Legal Services Corporations (LSC) and disability organizations on major, systemic disability rights issues involving the Individuals with Disabilities Education Act (IDEA), Americans with Disabilities Act (ADA), and the federal Medicaid Act. Recently in November 2014, Jim retired.
Founded in 2001, SDLC is a non-profit legal services organization dedicated to protecting and advancing the legal rights of people with disabilities throughout the South. It partners with the Southern Poverty Law Center, Protection and Advocacy (P&A) programs, Legal Services Corporations (LSC) and disability organizations on major, systemic disability rights issues involving the Individuals with Disabilities Education Act (IDEA), Americans with Disabilities Act (ADA), and the federal Medicaid Act. Recently in November 2014, Jim retired.