contributed by Dorothea Lantz, mom to Hunter (living with PWS) When Hunter was diagnosed with Prader-Willi syndrome as a baby, my world shifted overnight. Like so many parents in this community, I suddenly found myself learning a whole new language—growth hormone, hypotonia, food security, behavioral supports, specialists, therapies. I was trying to understand what life...
Ask Nurse Lynn: PWS and Procreation Possibilities
Question: Female, 19 years old, unknown subtype I have PWS and I’m wondering if I can have kids of my own. Nurse Lynn’s Response: Yes, it is possible, but it is rare. Most girls and women with PWS have a condition called hypogonadism. This means your body may not make the hormones needed for normal...
In Their Corner: Stories from PWS Sibling Advocates
April 10th is National Sibling Day, and we’re honoring it by sharing stories from a few incredible sibling advocates in the PWS community. From taking their voices to Capitol Hill to organizing coffee shop and marathon fundraisers, these advocates open up about what sibling advocacy looks like in their lives and the advice they have...
Diving in to Volunteering
We at PWSA | USA are incredibly grateful to our volunteers. These wonderful volunteers show up in a variety of capacities to help us carry out our mission to “enhance the quality of life of and empower those affected by Prader-Willi syndrome.” Some are older parents well-versed in the intricacies of the PWS community. Some...
Ask Nurse Lynn: Cataplexy Evaluation
Question: Male, 1 year old, imprinting subtype 1 year old, non-deletion imprinting center defect- he has been having “episodes”. He will be playing and then lose all muscle control. Head bobbing, falls over, almost like a little drunk person. Which is not normal, he has extremely good muscle control, always on the move and in...
Soleno Therapeutics to Be Acquired by Neurocrine Biosciences: What This Means for the PWS Community
On Monday, April 6, 2026, PWSA | USA received a letter from our partners at Soleno Therapeutics sharing significant news: Soleno has agreed to be acquired by Neurocrine Biosciences. Soleno describes this as an exciting development, noting that the transaction “marks an exciting milestone for our company and represents the next step in our efforts...
Growing up with PWS: A Sibling’s Story
Contributed by Hannah Kellerman, sibling to Audrey who passed away from complications of PWS in 2024. I first heard the words Prader-Willi Syndrome when I was three years old. I didn’t understand what those words had meant at the time; I was just so excited to have a new baby sister. Now, eighteen years later,...
Ask Nurse Lynn: Behavioral Outbursts and Psychiatric Support
Question: Male, 26 years old, deletion subtype My son has struggled with severe behavioral outbursts now at a rate of about 3-4 times a year, where bystanders will call police and he will either be arrested or brought to psych eval. Always calms down, is sent home. Had been on risperidone at increasing amounts as...
A Message from Soleno Therapeutics: Reflecting on One Year of VYKAT XR
One year ago, on March 26, 2025, the Prader-Willi syndrome community reached a historic milestone. For the first time, a treatment, VYKAT™ XR (diazoxide choline), was approved by the FDA specifically for hyperphagia in individuals ages 4 and older living with PWS. For a community that has long understood hyperphagia as the most urgent and...
Recipes from the PWS Community
Egyptian Lentil Soup (Shorbet ‘Ads) *submitted by Sarah Kasaby, mom to Khaled, living with PWS Ok so this is my beloved Egyptian Lentil Soup (Shorbet ‘Ads). It is very famous in winter, especially when you want something quick, nutritious, and keeps your body (and belly) warm. It’s perfect for our PWS diet because you literally...
HUD Changes Eviction Notice Rules for HUD-Assisted Housing
*submitted by Lisa Graziano As shared by the CA State Council on Developmental Disabilities in March, 2026 the U.S. Department of Housing and Urban Development (HUD) issued a rule that changes how much notice tenants receive before eviction for unpaid rent in HUD-assisted housing. What You Need to Know In 2024, HUD created a rule...
Ask Nurse Lynn: Supplements for Infants with PWS
Question: Female, 5 months old, UPD subtype We are looking for guidance on supplements for infants with Prader-Willi syndrome, particularly for brain development. Our daughter is 5 months, and we are beginning conversations about whether supplements may be appropriate for her at this stage. We understand that Dr. Miller has published general supplement recommendations for...
Aardvark Therapeutics Shares Additional Details on Voluntary Pause of HERO Clinical Trial
Last month, we shared that Aardvark Therapeutics announced a voluntary pause in enrollment and dosing for the Phase 3 HERO clinical trial evaluating ARD-101 for hyperphagia in individuals with Prader-Willi syndrome (PWS). In a new update, Aardvark has provided additional details to help explain this decision and outline next steps. According to the announcement shared...
Ask Nurse Lynn: Seizure and Neurology Appointment
Question: Male, 20 years old, deletion subtype My son had a seizure last Saturday where he lost consciousness and was stiff in his body, he then jolted awake and went on to have full body jerking for 7-8 seconds. The whole seizure lasted under 2 minutes, but he took around an hour to be fully...
PWSA | USA Board of Directors Member Spotlight: Tina Ihlenfeld
Board of Directors Member: Tina Ihlenfeld, MBA Board Role: Treasurer Years on the Board: 2 What first connected you to the Prader-Willi syndrome community, and what inspired you to join the PWSA | USA Board of Directors? I first became connected to the Prader-Willi syndrome community through my work at Prader-Willi Homes in Wisconsin. In...
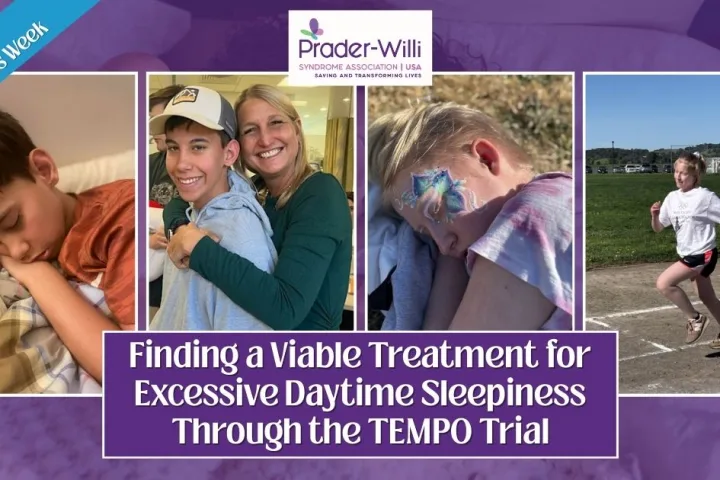
Finding a Viable Treatment for Excessive Daytime Sleepiness Through the TEMPO Trial
We are recognizing Sleep Awareness Week (March 9-14) and World Sleep Day (March 14) because disordered sleep is a common issue in individuals with Prader-Willi syndrome. Many of our loved ones are diagnosed with obstructive sleep apnea, central sleep apnea, cataplexy, narcolepsy, and/or excessive daytime sleepiness. These symptoms can greatly impact mental cognition, physical stamina,...
Ask Nurse Lynn: Dissociation and PWS Mental Health
Question: Female, 23 years old, deletion subtype Are there any disassociation or dissociative disorders in Prader- Willi? Is disassociation part of PWS? Nurse Lynn’s Response: Dissociation or a dissociative disorder is not considered a core or defining part of PWS. Experts who study PWS, including those summarized in the IPWSO Mental Health Network Report (June...
We’re Here: Then, Now, Always! Developmental Disabilities Awareness Month
March is Developmental Disabilities Awareness Month! Developmental disabilities are conditions that begin at birth or in childhood and may affect physical development, learning, language, or behavior. While these conditions can present challenges, they are only one part of a person’s story. Individuals with PWS are first and foremost people, with personalities, talents, preferences, humor, determination,...
Ask Nurse Lynn: Nutrition Guidelines and a Group Home
Question: Male, 38 years old, deletion subtype We are working with our son’s group home to help monitor his food intake. Is there a standard reduction in listed serving sizes we can recommend for them to follow to help staff with his meals? Nurse Lynn’s Response: I’m glad to hear that you’re working together with...
Important Update for the PWS Community: HERO Phase 3 Trial for ARD-101 Voluntarily Paused
We would like to share an important update from Aardvark Therapeutics. Aardvark has announced a voluntary pause of the Phase 3 HERO clinical trial, which is evaluating ARD‑101 for the treatment of hyperphagia in people with Prader‑Willi syndrome. The decision to pause the trial is based on reversible cardiac observations identified in a non-PWS, separate healthy...
Reflections from Rare Disease Week on Capitol Hill 2026
PWS advocates returned from EveryLife Foundation for Rare Diseases’s Rare Disease Week on Capitol Hill energized and inspired for the work ahead, especially as we look toward PWSA | USA’s D.C. Fly-In this May. Twenty-two PWS advocates, including four individuals living with Prader-Willi syndrome, gathered on Capitol Hill alongside 800+ other rare disease champions from...
Rare Disease Advocacy in the PWS Community
Rare Disease Advocacy in Orlando “On Rare Disease Day, I had the privilege of attending ‘Turning the Page on Prader‑Willi Syndrome’ in Winter Park, FL, to celebrate Dr. Destiny Pacha, an author, PWS education specialist, a friend and a true life‑changer for so many families in the PWS community. Hearing the story that began with...
NEW PWS Resource Available: Swallowing in PWS
We’re excited to share a new family-friendly guide: “Swallowing in Prader-Willi Syndrome.” This easy-to-understand resource helps parents and caregivers recognize signs of swallowing difficulties, understand silent aspiration, and learn practical mealtime and pill-swallowing tips to help keep children safe and healthy. From knowing when to ask for a swallow study to simple habits that can make...
Ask Nurse Lynn: Cortisol Levels, Testing, and Treatment
Question: Male, 2.5 years old, deletion subtype What are the implications for low cortisol levels in toddler boys with deletion subtype (type 2)? We’ve noticed some more fatigue and he sleeps longer during the night. What can and should be done about it? Our pediatrician ran a blood panel and cortisol came back at 3.7...
























 Perry A. Zirkel has written more than 1,500 publications on various aspects of school law, with an emphasis on legal issues in special education. He writes a regular column for NAESP’s Principal magazine and NASP’s Communiqué newsletter, and he did so previously for Phi Delta Kappan and Teaching Exceptional Children.
Perry A. Zirkel has written more than 1,500 publications on various aspects of school law, with an emphasis on legal issues in special education. He writes a regular column for NAESP’s Principal magazine and NASP’s Communiqué newsletter, and he did so previously for Phi Delta Kappan and Teaching Exceptional Children. Jennifer Bolander has been serving as a Special Education Specialist for PWSA (USA) since October of 2015. She is a graduate of John Carroll University and lives in Ohio with her husband Brad and daughters Kate (17), and Sophia (13) who was born with PWS.
Jennifer Bolander has been serving as a Special Education Specialist for PWSA (USA) since October of 2015. She is a graduate of John Carroll University and lives in Ohio with her husband Brad and daughters Kate (17), and Sophia (13) who was born with PWS. Dr. Amy McTighe is the PWS Program Manager and Inpatient Teacher at the Center for Prader-Willi Syndrome at the Children’s Institute of Pittsburgh. She graduated from Duquesne University receiving her Bachelor’s and Master’s degree in Education with a focus on elementary education, special education, and language arts.
Dr. Amy McTighe is the PWS Program Manager and Inpatient Teacher at the Center for Prader-Willi Syndrome at the Children’s Institute of Pittsburgh. She graduated from Duquesne University receiving her Bachelor’s and Master’s degree in Education with a focus on elementary education, special education, and language arts. Evan has worked with the Prader-Willi Syndrome Association (USA) since 2007 primarily as a Crisis Intervention and Family Support Counselor. Evans works with parents and schools to foster strong collaborative relationships and appropriate educational environments for students with PWS.
Evan has worked with the Prader-Willi Syndrome Association (USA) since 2007 primarily as a Crisis Intervention and Family Support Counselor. Evans works with parents and schools to foster strong collaborative relationships and appropriate educational environments for students with PWS. Staci Zimmerman works for Prader-Willi Syndrome Association of Colorado as an Individualized Education Program (IEP) consultant. Staci collaborates with the PWS multi-disciplinary clinic at the Children’s Hospital in Denver supporting families and school districts around the United States with their child’s Individual Educational Plan.
Staci Zimmerman works for Prader-Willi Syndrome Association of Colorado as an Individualized Education Program (IEP) consultant. Staci collaborates with the PWS multi-disciplinary clinic at the Children’s Hospital in Denver supporting families and school districts around the United States with their child’s Individual Educational Plan. Founded in 2001, SDLC is a non-profit legal services organization dedicated to protecting and advancing the legal rights of people with disabilities throughout the South. It partners with the Southern Poverty Law Center, Protection and Advocacy (P&A) programs, Legal Services Corporations (LSC) and disability organizations on major, systemic disability rights issues involving the Individuals with Disabilities Education Act (IDEA), Americans with Disabilities Act (ADA), and the federal Medicaid Act. Recently in November 2014, Jim retired.
Founded in 2001, SDLC is a non-profit legal services organization dedicated to protecting and advancing the legal rights of people with disabilities throughout the South. It partners with the Southern Poverty Law Center, Protection and Advocacy (P&A) programs, Legal Services Corporations (LSC) and disability organizations on major, systemic disability rights issues involving the Individuals with Disabilities Education Act (IDEA), Americans with Disabilities Act (ADA), and the federal Medicaid Act. Recently in November 2014, Jim retired.